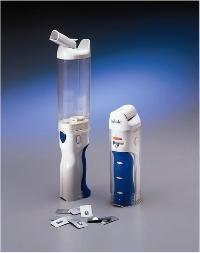
Exubera is now well on its way in Europe as well as the US.
The inhaler delivers insulin in dry form through an aerosol spray to the lung. The principle underlying inhaled insulin is that the large surface area of the lung enables the relatively large insulin molecules to cross from the lung and thence into the blood stream.
After inhalation, A relatively small amount of the dose inhaled is available for absorption (approximately 15%), but given a sufficient inhaled dose, this is clinically effective.
The onset of action is similar those of analogue insulin after s/c injections, with a duration of action which is between analogue and soluble insulin. Exubera will best suit people on multiple daily injections, who are having problems with injections.
Exubera will not be availible for people with severe asthma, those who smoke, people with lung disease or women of childbareing age. Some people may still need insulin injections. The product will be very closely watched, as it is a true medical breakthrough.
Pfizer and Sanofi are developing Exubera with Nektar Therapeutics, a small U.S. company that invented the insulin formulation and the inhaler for delivering it to the lungs.
Insider expects Pfizer to buy its partner Sanofi out of the Exubera project, once it has won a green light from regulators.
Source: Reuters, Ben Hirschler
No comments:
Post a Comment